The PSMA & Beyond 2025 Conference returned for its highly anticipated second edition, this time hosted in vibrant Los Angeles, California. Organized by University of California, San Francisco (UCSF), University of California, Los Angeles (UCLA), and the Prostate Cancer Foundation (PCF), this two-day event convened many of the world’s leading voices in prostate cancer theranostics, a cancer treatment approach that combines diagnostic imaging and targeted therapy. This highly specialized, niche conference drew over 600 attendees both virtually and in-person from around the globe, bringing together leaders at the forefront of cancer imaging and targeted therapy.
This multidisciplinary event featured stakeholders across molecular imaging, medical oncology, urology, radiation oncology, and other key disciplines—offering key insights to the rapidly advancing field of prostate-specific membrane antigen (PSMA) theranostics and precision medicine.
A Dynamic Program at the Cutting Edge
This year’s program explored both the current landscape and emerging directions in PSMA PET imaging and radioligand therapy. Sessions spanned a diverse range of topics—from the evolving role of bone scans, MRI, and PSMA PET in clinical decision-making, to therapeutic advances involving beta and alpha-emitting agents, radiopharmaceutical development, resistance mechanisms, and the emergence of novel tracers and ligands.
The conference also featured groundbreaking research in radiation measurement and the integration of artificial intelligence (AI), both aimed at enhancing the efficiency, accuracy, and personalization of theranostic approaches. Highlights included interactive tumor boards, engaging panel discussions, and visionary presentations that pushed the boundaries of what’s possible—not just in prostate cancer, but in theranostics more broadly.
In an exciting and timely development, Day 1 of the conference coincided with the U.S. Food and Drug Administration (FDA)’s announcement of an expanded indication for Pluvicto—the first FDA approved PSMA targeted theranostic which is now also approved earlier in the disease trajectory, prior to taxane-based chemotherapy. This marks a major milestone in expanding access and optimizing treatment sequencing for patients with metastatic prostate cancer.
Spotlight on WCM Rising Research Talent
The conference also hosted a vibrant poster session, featuring cutting-edge abstracts. Among the standout contributions was research by Dr. Valentina Marulanda-Corzo, Molecular Imaging and Therapeutics Research Fellow at WCM. Under the mentorship of Dr. Joseph R. Osborne, Chief of Molecular Imaging and Therapeutics, Dr. Marulanda-Corzo presented her abstract titled “PSMA-Targeted Actinium-225 Therapy in Metastatic Castration-Resistant Prostate Cancer: Integrating Imaging Biomarkers with Clinical Outcomes in Monotherapy and Combination Therapy.”
Her work showcased how quantitative imaging biomarkers can provide insights that go beyond PSMA expression, offering critical prognostic and therapeutic value. By integrating imaging data with clinical outcomes, her research underscores the pivotal role of imaging in guiding personalized treatment strategies and advancing the field of precision oncology.
Selected as one of only ten “Power Pitch” presentations, Dr. Marulanda-Corzo’s abstract was ultimately recognized as one of the top three poster presentations of the entire conference—an incredible achievement and a powerful testament to the innovation and leadership emerging from Weill Cornell Medicine.
Weill Cornell Medicine at the Forefront

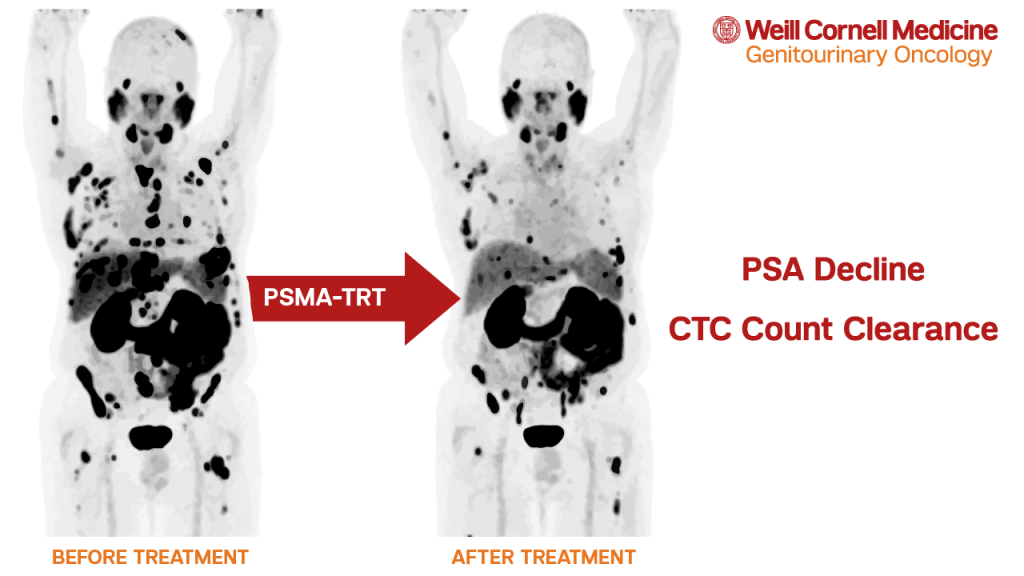
Weill Cornell Medicine (WCM) was proudly represented at this premier conference. Dr. Scott Tagawa, a medical oncologist at WCM and an internationally recognized leader in PSMA-targeted therapy, delivered a compelling presentation titled “The Role of PSMA Targeted Antibodies.” Drawing from ongoing clinical trials at WCM, Dr. Tagawa explored the development and therapeutic promise of J591, a monoclonal antibody that targets the extracellular domain of PSMA—enabling highly selective delivery of both beta and alpha-emitting radionuclides.
His presentation emphasized the clinical relevance of this platform, particularly in patients with low or varying PSMA expression, where small-molecule ligands may fall short. He shared promising clinical outcomes using Actinium-225-labeled J591, and highlighted how alpha emitters are reshaping the therapeutic landscape.
Notably, Dr. Tagawa explored the synergistic potential of combination strategies—such as pairing alpha and beta emitters or integrating alpha therapy with immunotherapy—to deepen response and overcome treatment resistance. His talk reinforced that the future of PSMA-targeted therapy lies not only in precision, but in innovation, through multidimensional approaches that could redefine cancer care.
This year’s PSMA & Beyond Conference served as a powerful reminder that the fusion of imaging and therapy is not just a scientific frontier—it is a transformative movement redefining how we approach cancer care. At Weill Cornell Medicine, we are proud to be at the center of this evolution, advancing theranostics through research, collaboration, and a deep commitment to patient-centered innovation.