Radiopharmaceuticals (also sometimes called targeted radionuclide therapy or similar) are a type of medication that uses particles of radiation injected into the bloodstream to affect cancer. Radiopharmaceuticals are typically comprised of a radioactive particle, known as a radionuclide, sometimes combined with a targeting molecule that directs the radiation or points it towards a particular place. When injected into the body, this type of therapy is able to deliver radiation directly to the pointed target, limiting the exposure to the surrounding healthy tissue.
In prostate cancer, radiopharmaceuticals can be used in patients with metastatic prostate cancer that is not responding to hormonal therapy. While these treatments aren’t a cure, they are able to help these patients live longer with a better quality of life.
There are currently two radiopharmaceuticals approved by the U.S. Food and Drug Administration (FDA) for the treatment of metastatic prostate cancer. Both of these treatments are given via an intravenous (IV) injection or infusion, similar to other forms of cancer treatment.
Radium Ra 223 dichloride (Xofigo) was approved in 2013 and is used to target bone. In the setting of prostate cancer that has metastasized to the bones, patients live longer with this treatment.
Lutetium Lu 177 vipivotide tetraxetan (aka 177Lu-PSMA-617, Pluvicto) was approved in 2022 and delivers prostate-specific membrane antigen (PSMA) targeted radiation to prostate cancer cells that express PSMA (which encompasses the majority of prostate cancer). It can be used to treat PSMA-expressing cancer that has metastasized anywhere in the body, including the bones, internal organs, and lymph nodes.
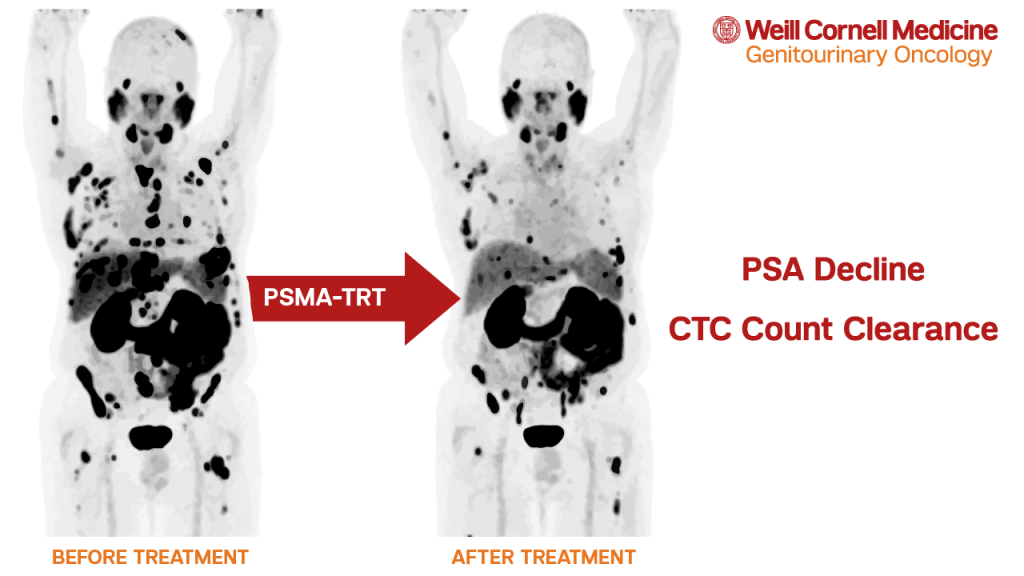
New radiopharmaceutical treatment options are also being evaluated in clinical trials. A phase 1 clinical trial led by Dr. Scott Tagawa at Weill Cornell Medicine/NewYork-Presbyterian Hospital (WCM/NYP) evaluated the radiopharmaceutical 225AC-J591 in patients with advanced prostate cancer. Multiple additional studies of 225AC-J591 alone or in combination with other therapies are now underway at WCM to further assess the safety and efficacy of 225AC-J591.
In addition, multiple additional radiopharmaceuticals are in development, including drugs such as 67Cu-SAR-bisPSMA, 177Lu-rhPSMA-10.1, and TLX591. A full list of our open clinical trials in this area can be found here.
Patients with prostate cancer interested in radiopharmaceutical treatment should speak to their doctor to determine if this therapy may be a good option based on their individual situation. Patients will need to undergo two scans to determine if they are eligible to receive a radiopharmaceutical. One may be a bone scan to determine if the cancer has metastasized to the bones and the second may be a PSMA positron emission tomography (PET) scan to determine if the cancer cells have a high level of PSMA protein.
To learn more about radiopharmaceuticals for metastatic prostate cancer, read this in-depth patient-friendly overview from Everyday Health featuring insights from Dr. Scott Tagawa, facts about radiopharmaceuticals, questions to ask your doctor about this treatment option, and more.
You can also learn more about the prostate clinical trials open at Weill Cornell Medicine/NewYork-Presbyterian Hospital here.